(Image source from: health.economictimes.indiatimes.com)
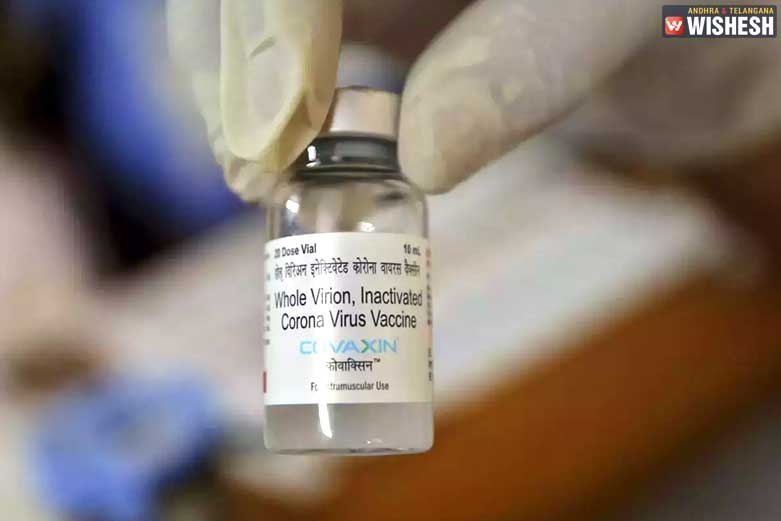
Expert panel approves Bharat Biotech's children's vaccine for trials:- Bharat Biotech's Covaxin has been recommended by the expert panel for the second and third phase of clinical trials for the children aged between 2 and 18 years. The Subject Expert Committee (SEC) has granted the permission to conduct the phase 2,3 clinical trials on the children. 525 students are selected across the country and the trials will take place at AIIMS New Delhi, AIIMS Patna and Nagpur's Meditrina Institute of Medical Sciences as per the reports. The Centre informed Supreme Court that the Indian government provided no financial aide for the research or the development of Covaxin and Covishield in the country.
The Expert Committee of Central Drugs Standard Control Organization (CDSCO) deliberated upon the application seeking the permission about the clinical trials. The safety, immunogenicity and reactogenicity of Covaxin will be verified among the children during the trials. Several meetings are conducted by the Subject Expert Committee through a meeting that was dated February 24th. Bharat Biotech was asked to submit a complete a revised clinical trial protocol. Covaxin is developed by Bharat Biotech in association with the Indian Council of Medical Research (ICMR). The clinical trials are expected to be completed this year and the vaccination would be available for children early next year if things go as per the plan.